Stem Cell Therapy to Cure Erectile Dysfunction
The Blue Cells will provide the first curative treatment for severe erectile dysfunction on the market
What are Blue Cells?
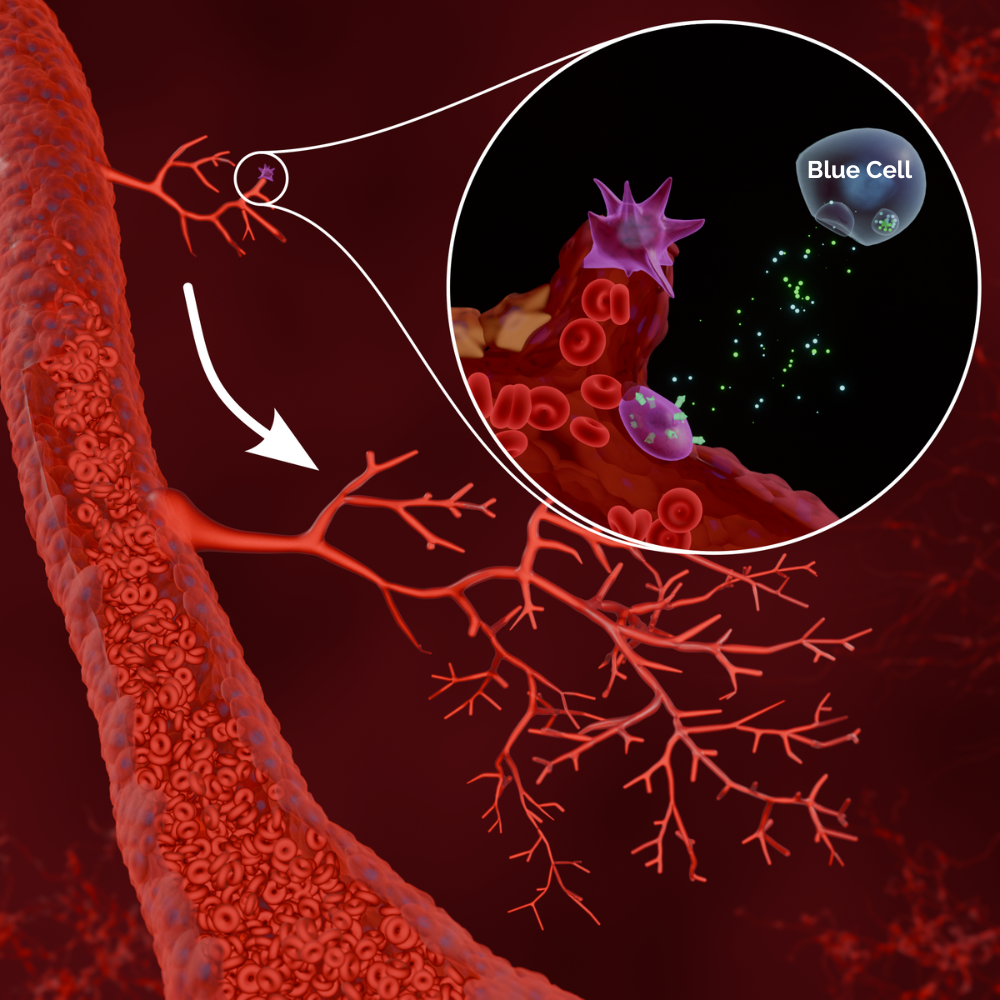
Blue Cells represent an off-the-shelf allogeneic stem-cell platform engineered to trigger blood-vessel growth and neural repair, offering a curative strategy for difficult-to-treat disorders. By secreting potent angiogenic mediators like VEGF alongside essential neurotrophic factors, Blue Cells foster vascular regeneration and nerve restoration. Our flagship candidate, BlueC-231, targets severe erectile dysfunction (ED).
Erectile Dysfunction
Erectile dysfunction impacts over 85 million men globally, with approximately 30 percent unresponsive to existing drug treatments.
Today, no curative therapies exist.
Men treated with radical prostatectomy for prostate cancer face the highest risk, as the surgery often injures nerves essential for an erection. Each year, about 490,000 prostatectomies are performed in the Western world, and the majority of these patients continue to experience severe ED one year later.
Cell therapy for erectile dysfunction
Blue Cell has developed a scalable, safe and potentially curative solution for patients suffering from severe ED.
Preliminary research examining the potential of stem-cell treatment for ED has proven highly encouraging.
In addition, preclinical in vitro and in vivo data have shown that angiogenesis and regrowth of endothelial and nerve cells achieved after treatment with allogeneic stem cells are superior to those achieved with autologous stem cells.
What are Blue Cells?

Blue Cells are an allogeneic (‘off-the-shelf’) stem cell therapy product that work by promoting angiogenesis (the development of new blood vessels), repairing the endothelium (the inner lining of blood vessels), vascular smooth muscle, and neurons – but also by inhibiting inflammation.
Blue Cells will provide a therapeutic treatment for post prostatectomy patients suffering from severe erectile dysfunction.
Erectile Dysfunction

Erectile Dysfunction is a medical condition currently estimated to affect more than 85 million men worldwide, and 30% of them do not respond to drug therapy.
Currently, no curative treatment is available for erectile dysfunction.
Among those most seriously affected by ED are men who must undergo a radical prostatectomy – removal of the prostate gland and surrounding tissue – due to prostate cancer. This type of surgery can damage nerves in the penis.
After prostatectomy, ED can be long-lasting. Most of the 490,000 men in the western world who undergo major prostate cancer surgery each year still suffer severe ED, a year after their operation.
Cell therapy for erectile dysfunction

Blue Cell has developed a scalable, safe and potentially curative solution for patients suffering from severe ED.
Preliminary research examining the potential of stem cell treatment for ED has proved to be highly encouraging.
In addition, preclinical in vitro and in vivo data have shown angiogenesis and endothelial cell and nerve cell regrowth achieved after treatment with allogeneic stem cells to be superior to that achieved after treatment with autologous stem cells.

About Blue Cell Therapeutics
At Blue Cell Therapeutics, our mission is to revolutionise the treatment of erectile dysfunction, using Blue Cell therapy, for men with post-(radical) prostatectomy. We are committed to developing a curative, ‘off-the-shelf’ allogeneic stem cell therapy for severe erectile dysfunction with enhanced safety, efficacy, and accessibility.

Our Team
Blue Cell Therapeutics was founded by Søren P Sheikh MD, PhD, HD, after he demonstrated the curative potential of adipose-derived stem cells for ED in an autologous phase I clinical trial. Since then, Blue Cell Therapeutics has developed standardised methods to isolate and culture stem cells, while maintaining or even enhancing their biological effects.
Blue Cell Therapeutics has gathered a high profile management team and a skilled, dedicated team of scientific employees.



